|
On the other hand, the extremely high speed of electrons (unlike that of planets) suggested that relativity be brought into the picture. When atoms absorb energy, the electrons at a particular level are pushed up to higher levels (at bigger. On the one hand, the positive analogy suggested that electrons (like planets) revolved around the center of mass of the atom (solar system). Bohrs solar system model of the atom is the way that most people think about atoms today. Bohrs model was not perfect and was soon superseded by the more accurate Schrödinger model, but it was sufficient to evaporate any remaining. It described the overall structure of the atom, how atoms bond to each other, and predicted the spectral lines of hydrogen. Furthermore, a related analogy between electrons and planets played a significant role in Bohr’s subsequent articulation of the model. The Bohr model of the atom was the first complete physical model of the atom. Bohr proposed that electrons do not radiate energy as they orbit the nucleus, but exist. The electron is not allowed to occupy any of the spaces in between the orbits. The elaboration of the negative analogy led to the conclusion that the electron could move only in certain discrete orbits and its energy and angular momentum were accordingly restricted. According to the Bohr model, or planetary model, the electrons encircle the nucleus of the atom in specific allowable paths called orbits. 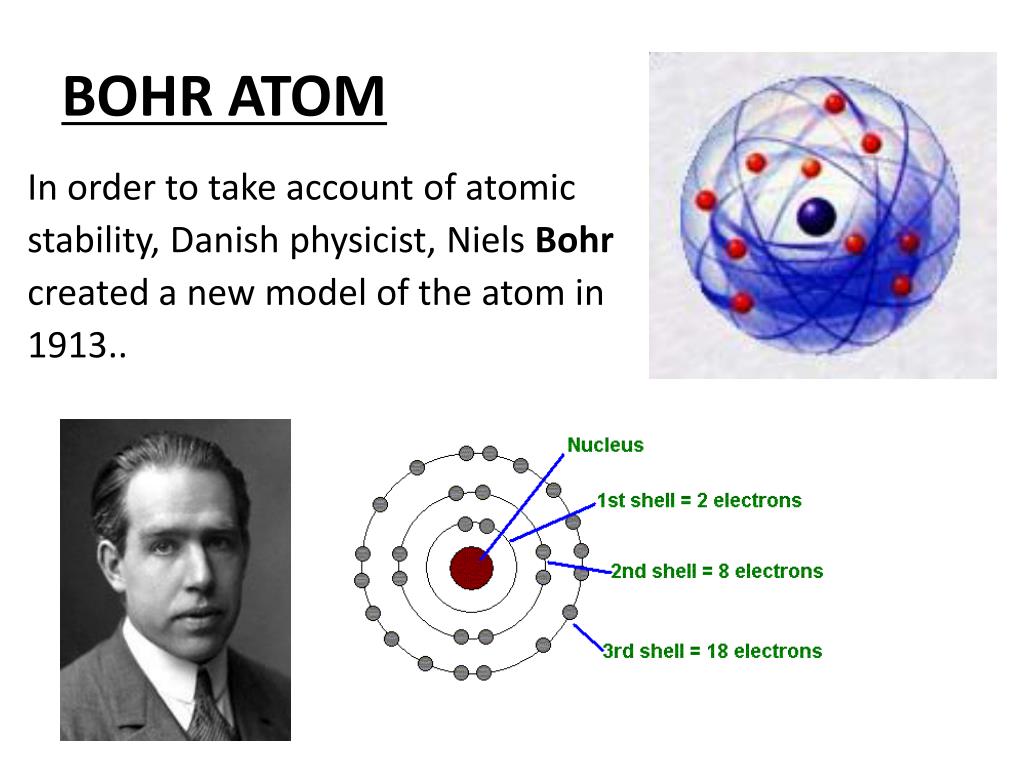 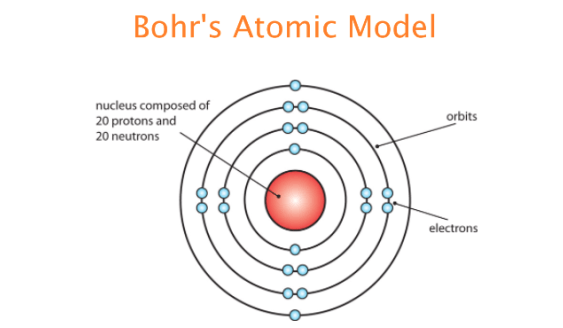
We will argue that it was the negative, rather than the positive, analogy between intra-atomic electrons and the rings of Saturn that motivated Bohr to adopt and develop Rutherford’s atomic model. In our paper we will stress the significance of negative analogies for the development of Bohr’s atom. The electronic structure explains the location and movement of the electrons in atoms. However, she underappreciated the role of negative analogies in model-building. The electrons exist in a cloud orbiting the nucleus. A Bohr model of a chlorine atom shows a nucleus surrounded by three concentric rings. Every model sacrifices some accuracy for simplicity, visibility, or usability. However, its important to remember that no scientific model is perfect. They also help us explain and predict the behavior of atoms. Bohr model of an atom consists of a small nucleus that contains protons and neutrons, this nucleus is surrounded by different electron shells or energy levels where electrons are revolved in a definite circular path similar to the structure of. Models help us visualize atomic structure. (b) The energy of the orbit becomes increasingly less negative with increasing n. 2 The Bohr Model of the Hydrogen Atom (a) The distance of the orbit from the nucleus increases with increasing n. Modern scholarship on analogy takes its departure from the work of Mary Hesse, who pointed out the existence of negative analogies between two different physical systems, that is, those respects in which the two systems clearly differ. Bohr model or RutherfordBohr diagram, presented by Niels Bohr and Ernest Rutherford in 1913. In this state the radius of the orbit is also infinite. The significant role of models and analogies in scientific practice has been widely recognized.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
